Why O-Rings Crack
We’ve all heard of the ozone layer and its effects on humans and the environment. But it also affects materials, such as O-rings.
The good news is that it mostly affects O-rings made out of nitrile rubber, also known as buna or buna-n. The bad news is that they can be affected by ultraviolet light, electrical arcing, and electromagnetic fields.
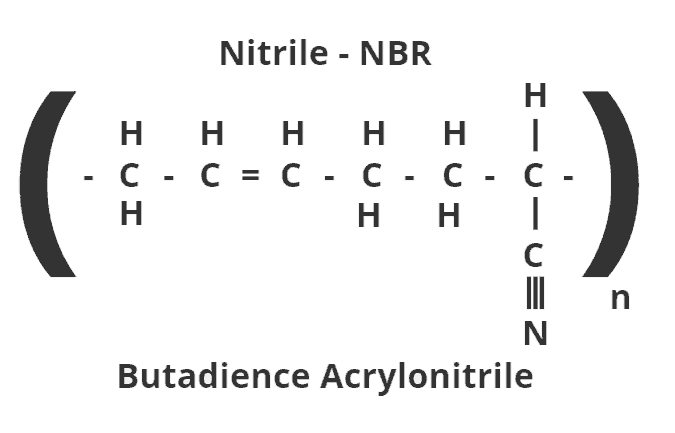
Here’s why. Nitrile rubber is a polymer — the word is derived from the Greek which means many units. Each molecule consists of long chains, of individual units made up of carbon, hydrogen, and nitrogen linked by single, double or triple bonds.
The double bond between the second and third atom is a weak spot which is vulnerable to oxygen atoms found in ozone. Exposure to oxygen atoms cuts the polymer chain — which creates a crack in the O-ring. The longer the exposure to ozone, the more the crack will deepen and more cracks will appear.
How can ozone cracking be prevented?
Storing O-rings correctly will go a long way to preventing cracking.
Install within 24 hours
Once you’ve installed a nitrile O-ring on a fitting, make sure the mating part is installed with 24 hours. Greasing O-rings before assembly will provide protection against ozone.
Depending on your application, nitrile rubber can be an excellent solution. It’s widely used due to its resistance to oils, fuels, and other chemicals in a wide variety of industries. However, if you anticipate long-term exposure to ozone, it’s best to use an ozone-resistant material such as HNBR, EPDM, or fluorocarbon.
Talk to our experts about the material best suited to your application.
>> Reference: http://blog.parker.com/why-does-my-o-ring-have-all-these-cracks-in-it




